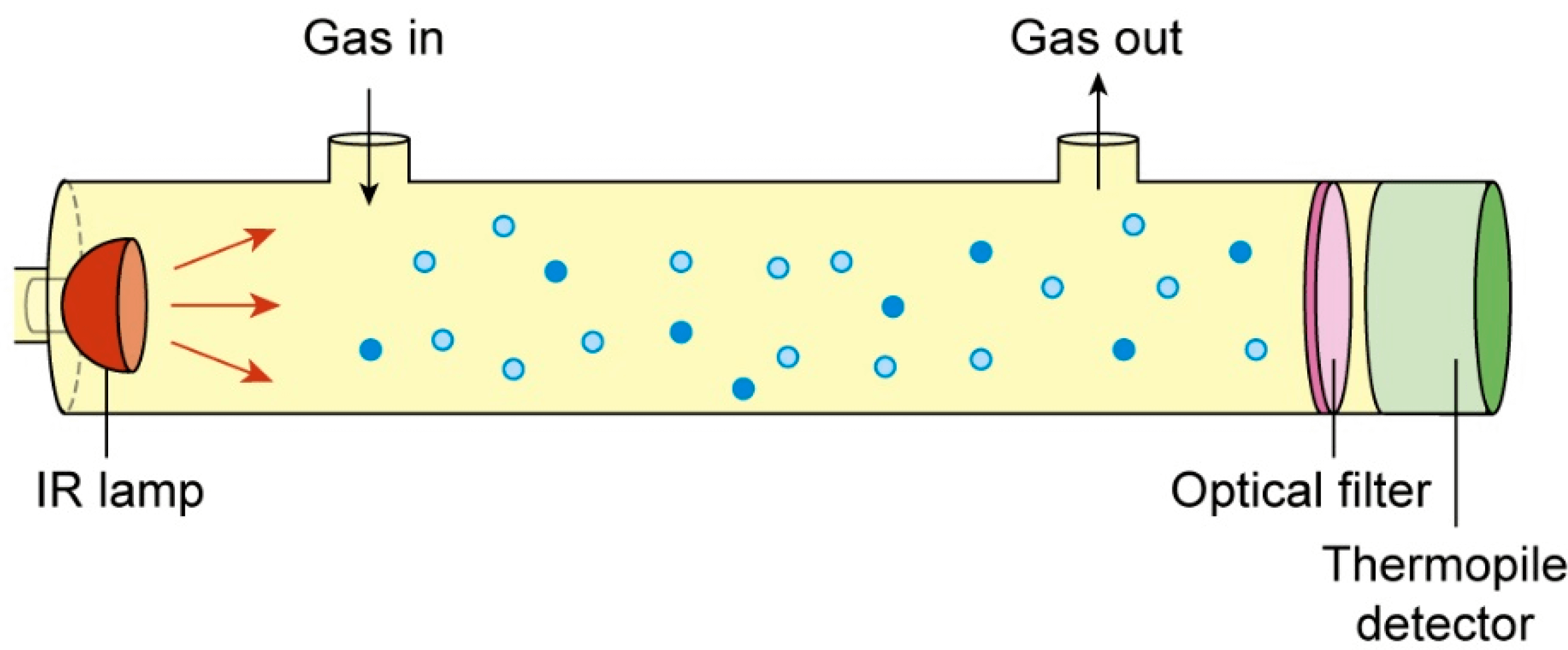
When working with gases, it's crucial to understand their properties and exposure limits to ensure safety and prevent potential health hazards. Gas exposure limits provide guidelines for the maximum allowable concentration of a gas in the air that workers can be exposed to without adverse effects. These limits vary depending on the specific gas and its properties, such as toxicity, flammability, and reactivity.
Introduction to Exposure Safety
In this article, we will explore the key aspects of gas exposure limits and gas properties to help you make informed decisions when dealing with gases in your workplace. We used various sources to populate our tables, which included source data from:
OSHA Data
https://www.osha.gov/annotated-pels/table-z-1
NIOSH Data
https://www.cdc.gov/niosh/npg/
ACGIH Data
https://www.acgih.org/
WHO Data
https://www.ncbi.nlm.nih.gov/books/NBK574591/table/ch3.tab26/
EPA Data
https://www.epa.gov/criteria-air-pollutants/naaqs-table
Acrylonitrile (C3H3N)
Acrylonitrile is a colorless, flammable, and toxic liquid with a pungent odor. It is used in the production of plastics, synthetic fibers, and rubber. Exposure to acrylonitrile can occur through inhalation, skin contact, or ingestion, and it is classified as a probable human carcinogen by the EPA and IARC.
Acrylonitrile Fact Sheet.
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 2 ppm average over 8 hours
Ceiling: 10ppm average over 15 minutes
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 1 ppm average over 10 hours
Ceiling: 10ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
2 ppm average over 8 hours
|
Acetylene (C2H2)
Acetylene (C2H2) is a colorless, flammable gas with a garlic-like odor. It is the simplest alkyne, consisting of two carbon atoms triple-bonded together with one hydrogen atom bonded to each carbon. Acetylene is used in welding and cutting torches, and as a raw material for various chemical syntheses.
Acetylene Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 2500 ppm average over 8 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
NA
|
Ammonia (NH3)
Ammonia is a colorless gas with a pungent odor. It is composed of nitrogen and hydrogen (NH3). Ammonia is used in fertilizers, refrigerants, and cleaning products. Exposure to high concentrations can cause eye, nose, and throat irritation, and in severe cases, lung damage or death. Ammonia is flammable and explosive at high concentrations.
Ammonia Fact Sheet.
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 50 ppm average over 8 hours
|
| National Institute for Occupational Safety & Health (NIOSH) |
REL: 25 ppm average over 10 hours
Ceiling: 35ppm average over 15 minutes |
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV:
25 ppm average over 8 hours |
Arsine (AsH3)
Arsine (AsH3) is a colorless, flammable, and highly toxic gas with a garlic-like odor. It is the simplest arsenic hydride, consisting of one arsenic atom bonded to three hydrogen atoms. Arsine is used in the semiconductor industry and is a byproduct of some industrial processes. Exposure can be fatal.
Arsine Fact Sheet
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 0.05 ppm average over 8 hours
Ceiling: 0.002 mg/m3 average over 15 minutes |
| National Institute for Occupational Safety & Health (NIOSH) |
REL: 0.0006 ppm never exceed this limit
|
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV:
0.005 ppm average over 8 hours |
Benzene (C6H6)
Benzene (C6H6) is a colorless, flammable liquid with a sweet odor. It is an aromatic hydrocarbon consisting of a ring of six carbon atoms, each bonded to one hydrogen atom. Benzene is a key component in the production of various chemicals, plastics, and pesticides, but is also a known carcinogen.
Benzene Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1 ppm average over 8 hours
Ceiling: 5ppm average over 15 minutes |
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
Ceiling: 1ppm average over 15 minutes |
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
0.5 ppm average over 8 hours
|
Bromine (Br2)
Bromine (Br) is a reddish-brown liquid at room temperature with a strong, unpleasant odor. It is a halogen element and the only liquid nonmetallic element. Bromine is used in the production of flame retardants, water treatment chemicals, and pharmaceuticals. It is toxic and can cause skin and respiratory irritation.
Bromine Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
Ceiling: 0.3ppm average over 15 minutes |
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
0.1 ppm average over 8 hours
|
Carbon Disulfide (CS₂)
Carbon disulfide (CS2) is a colorless, volatile liquid with a sweet, ethereal odor. It consists of one carbon atom double-bonded to two sulfur atoms. Carbon disulfide is used as a solvent and in the production of rayon, cellophane, and pesticides. It is highly flammable and toxic, affecting the nervous system.
Carbon Disulfide Fact Sheet
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 20 ppm average over 8 hours
Ceiling: 30 ppm average over 15 minutes |
| National Institute for Occupational Safety & Health (NIOSH) |
REL: 1 ppm average over 10 hours
Ceiling: 10 ppm average over 15 minutes |
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV:
1 ppm average over 8 hours |
Carbon Monoxide (CO)
Carbon monoxide (CO) is a colorless, odorless, and tasteless gas. It is formed by the incomplete combustion of carbon-containing fuels. Carbon monoxide binds strongly to hemoglobin in red blood cells, reducing oxygen delivery to the body. Exposure to high levels can cause headaches, dizziness, confusion, and death by asphyxiation.
Carbon Monoxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 50 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 35 ppm average over 10 hours
Ceiling: 200 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
25 ppm average over 8 hours
|
|
World Health Organization (WHO)
|
AQG: 4 µg/m3 average over 24 hours |
|
Environmental Protection Agency (EPA)
|
NAAQS: 9 ppm average over 1 hour
|
Chlorine (Cl2)
Chlorine (Cl) is a yellowish-green gas at room temperature with a pungent, irritating odor. It is a halogen element and a strong oxidizing agent. Chlorine is used in water treatment, as a disinfectant, and in the production of various chemicals and plastics. Exposure can cause respiratory issues and skin irritation.
Chlorine Fact Sheet
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 1 ppm not to be exceeded
|
| National Institute for Occupational Safety & Health (NIOSH) |
Ceiling: 0.5 ppm average over 15 minutes |
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV:
0.5 ppm average over 8 hours
STEL: 1 ppm average over 15 minute
|
Chlorine Dioxide (ClO₂)
Chlorine dioxide is a pale yellow gas with a pungent odor, used as a bleaching agent and disinfectant. It is a powerful oxidizer, effective against bacteria, viruses, and other microorganisms. In water treatment, it eliminates taste and odor compounds. However, it can be toxic if mishandled or inhaled.
Chlorine Dioxide Fact Sheet
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 0.1 ppm average over 8 hours
|
| National Institute for Occupational Safety & Health (NIOSH) |
REL: 0.1 ppm average over 10 hours |
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV: 0.1 ppm average over 8 hours
STEL: 0.3 ppm average over 15 minute
|
Cyclohexanol (C6H12O)
Cyclohexanol is a colorless, viscous liquid with a mild, sweet odor. It is an organic compound with the formula C6H11OH, consisting of a cyclohexane ring with a hydroxyl group attached. Cyclohexanol is used as a solvent and in the production of plastics, detergents, and pharmaceuticals. It is slightly soluble in water.
Cyclohexanol Fact Sheet
Agency
|
Limits
|
| Occupational Safety & Health Administration (OSHA) |
PEL: 50 ppm average over 8 hours
|
| National Institute for Occupational Safety & Health (NIOSH) |
REL: 50 ppm average over 10 hours
|
| American Conference of Government Industrial Hygienists (ACGIH) |
TLV: 50 ppm average over 8 hours |
Diborane (B2H6)
Diborane is a colorless, highly reactive, and flammable gas with the formula B2H6. It has a repulsive odor and is lighter than air. Diborane is used as a reducing agent, rubber vulcanizer, and catalyst in organic synthesis. It is toxic and can spontaneously ignite in air, requiring careful handling.
Diborane Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
0.1 ppm average over 8 hours
|
Ethyl alcohol (Ethanol C2H5OH)
Ethyl alcohol, also known as ethanol (C2H5OH), is a colorless, flammable liquid with a characteristic odor. It is the active ingredient in alcoholic beverages and is also used as a solvent, disinfectant, and fuel additive. Ethanol is produced by fermenting sugars from various sources, such as grains and fruits. It has a wide range of applications in industries like pharmaceuticals, cosmetics, and chemical synthesis.
Ethyl Alcohol Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1000 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 1000 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
1000 ppm average over 8 hours
|
Ethylene Oxide (C2H4O)
Ethylene oxide is a colorless, flammable gas with a sweet, ether-like odor. It has the formula C2H4O and is the simplest cyclic ether. Ethylene oxide is used in the production of ethylene glycol, polyester fabrics, and detergents. It is also a powerful sterilant for medical equipment, but it is toxic and a known carcinogen.
Ethylene Oxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1 ppm average over 8 hours
Ceiling: 5 ppm average over 15 minutes
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.5 ppm average over 10 hours
Ceiling: 5 ppm average over 10 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
1 ppm average over 8 hours
|
Formaldehyde (CH2O)
Formaldehyde is a colorless, flammable gas with a pungent odor. It has the formula CH2O and is the simplest aldehyde. Commonly used as a preservative in medical laboratories and mortuaries, formaldehyde is also found in household products like glues and pressed-wood. It is a known carcinogen and can cause irritation to the eyes, nose, and throat.
Formaldehyde Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.75 ppm average over 8 hours
Ceiling: 2 ppm average over 15 minutes
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.016 ppm average over 10 hours
Ceiling: 0.1 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
0.3 ppm average over 8 hours
|
Fluorine (F2)
Fluorine is a pale yellow, highly reactive, and corrosive gas with the symbol F. It is the lightest halogen and the most electronegative element. Fluorine readily forms compounds with most other elements, and it is used in the production of fluoropolymers, such as Teflon, and in the enrichment of uranium for nuclear fuel. Due to its reactivity, fluorine is extremely dangerous and must be handled with great care.
Fluorine Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
1 ppm average over 8 hours
STEL: 2 ppm average over 15 minute
|
Hexane (C6H14)
Hexane (C6H14) is a colorless, flammable liquid hydrocarbon with a gasoline-like odor. It is a non-polar solvent commonly used in industrial applications, such as oil extraction, cleaning agents, and adhesives. Hexane is also used in laboratories for chemical reactions and chromatography. Exposure to hexane can cause neurological damage and respiratory irritation.
Hexane Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 500 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 50 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 50 ppm average over 8 hours
|
Hydrazine (N2H4)
Hydrazine is a colorless, flammable liquid with the formula N2H4. It has a strong ammonia-like odor and is highly toxic. Hydrazine is used as a powerful reducing agent, a rocket propellant, and a blowing agent for plastics. It is also used in the production of pharmaceutical and agricultural chemicals. Hydrazine is carcinogenic and can cause severe burns, requiring extreme caution when handling.
Hydrazine Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.03 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
0.01 ppm average over 8 hours
|
Hydrogen Bromide (HBr)
Hydrogen bromide is a colorless, corrosive gas with a pungent odor, having the formula HBr. It is highly soluble in water, forming hydrobromic acid. Hydrogen bromide is used in the production of inorganic bromides, organic bromination reactions, and as a catalyst in industrial processes. It is toxic and can cause severe burns, requiring proper safety precautions when handling.
Hydrogen Bromide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 3 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 3 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
2 ppm average over 8 hours
|
Hydrogen Chloride (HCl)
Hydrogen chloride is a colorless, corrosive gas with a pungent odor and the formula HCl. It is highly soluble in water, forming hydrochloric acid. Hydrogen chloride is used in the production of vinyl chloride for PVC, in metal pickling, and in the oil and gas industry. It is toxic and can cause severe burns to the skin, eyes, and respiratory tract, necessitating proper safety measures during handling.
Hydrogen Chloride Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 5 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 5 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
2 ppm average over 8 hours
|
Hydrogen Cyanide (HCN)
Hydrogen cyanide is a colorless, highly poisonous gas or liquid with a faint, bitter almond-like odor. Its chemical formula is HCN. It is used in the production of plastics, synthetic fibers, and pesticides. Hydrogen cyanide inhibits cellular respiration, leading to rapid death. It was used as a chemical weapon and in Nazi gas chambers. Extreme caution is essential when handling this highly toxic substance.
Hydrogen Cyanide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 10 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 4.7 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
4.7 ppm average over 8 hours
|
Hydrogen Fluoride (HF)
Hydrogen fluoride is a colorless, corrosive gas or liquid with a strong, irritating odor. Its chemical formula is HF. It is highly soluble in water, forming hydrofluoric acid. Hydrogen fluoride is used in the production of fluoropolymers, fluorocarbons, and in the etching of glass and silicon wafers. It is extremely toxic and can cause severe burns, requiring specialized protective equipment and emergency procedures when handling.
Hydrogen Fluoride Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 3 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 3 ppm average over 10 hours
Ceiling: 6 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 0.5 ppm average over 8 hours
|
Hydrogen Peroxide (H2O2)
Hydrogen peroxide is a clear, colorless liquid with the formula H2O2. It is a strong oxidizer and is used as a bleaching agent, disinfectant, and in rocketry as a propellant. Household solutions are typically 3-6% concentration, while industrial grades can reach 90%. It decomposes into water and oxygen gas. Ingestion of concentrated hydrogen peroxide can be fatal, and contact with skin or eyes causes irritation.
Hydrogen Peroxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 1 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
1 ppm average over 8 hours
|
Hydrogen Sulfide (H₂S)
Hydrogen sulfide is a colorless, flammable, and highly toxic gas with a characteristic rotten egg odor. Its chemical formula is H2S. It occurs naturally in volcanic gases, hot springs, and from bacterial breakdown of organic matter. Hydrogen sulfide is used in the production of sulfuric acid and as an analytical reagent. Exposure to high concentrations can cause rapid unconsciousness and death, requiring immediate evacuation and proper protective equipment in industrial settings.
Hydrogen Sulfide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 20 ppm average over 8 hours
Ceiling: 50 ppm average over 15 minutes
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 10 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
1 ppm average over 8 hours
|
Isopropyl alcohol (IPA Propanol C3H8O)
Isopropyl alcohol, also known as rubbing alcohol, is a colorless, flammable liquid with a strong odor. It is commonly used as a disinfectant and solvent. With a chemical formula of C3H8O, it is often found in concentrations of 70-99%. Isopropyl alcohol has various applications in healthcare, cleaning, and industrial settings.
Isopropyl Alcohol Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 400 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 400 ppm average over 10 hours
Ceiling: 500 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
200 ppm average over 8 hours
STEL: 400 ppm average over 15 minutes
|
Methyl alcohol (Methanol CH₃OH)
Methyl alcohol, also known as methanol (CH3OH), is a colorless, flammable liquid with a slightly sweet odor. It is the simplest alcohol and is commonly used as a solvent, antifreeze, and fuel additive. However, methanol is highly toxic if ingested, inhaled, or absorbed through the skin, and can cause severe health issues, including blindness and death.
Methyl Alcohol Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 200 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 200 ppm average over 10 hours
Ceiling: 250 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
200 ppm average over 8 hours
STEL: 250 ppm average over 15 minutes
|
Nitric Oxide (NO)
Nitric oxide is a colorless, toxic gas with the formula NO. It is a free radical and is produced naturally in the body as a signaling molecule. Nitric oxide plays a crucial role in regulating blood pressure, neurotransmission, and immune response. Industrially, it is used in the production of nitric acid and as an intermediate in the synthesis of other chemicals. At high concentrations, nitric oxide can cause respiratory irritation and damage.
Nitric Oxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 25 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 25 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
25 ppm average over 8 hours
|
Nitrogen Dioxide (NO₂)
Nitrogen dioxide is a reddish-brown, highly reactive gas with a pungent, acrid odor. Its chemical formula is NO2. It is formed during high-temperature combustion processes, such as in vehicle engines and power plants. Nitrogen dioxide is a major component of smog and contributes to acid rain. Exposure can cause respiratory irritation, reduced lung function, and increased susceptibility to infections. It is also used in the production of nitric acid and as an oxidizing agent in various industrial processes.
Nitrogen Dioxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 5 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 1 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
OEL: 3 ppm
average over 8 hours
STEL: 5 ppm average over 15 minutes |
|
World Health Organization (WHO)
|
AQG: 25 µg/m3 average over 8 hours
|
|
Environmental Protection Agency (EPA)
|
NAAQS: 100 ppb average over 1 hour
|
Ozone (O₃)
Ozone is a pale blue gas with a distinctive odor, composed of three oxygen atoms (O3). It occurs naturally in the upper atmosphere, forming the ozone layer that protects Earth from harmful UV radiation. At ground level, ozone is a major component of smog, formed by reactions between nitrogen oxides and volatile organic compounds in the presence of sunlight. Ozone is a powerful oxidant and can cause respiratory irritation, aggravate asthma, and damage crops. It is also used in water treatment and as a disinfectant.
Ozone Fact Check
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
|
|
World Health Organization (WHO)
|
AQG: 100 µg/m3 average over 8 hours
|
|
Environmental Protection Agency (EPA)
|
NAAQS: 0.70 ppm average over 8 hours
|
Oxygen (O₂)
Oxygen is a colorless, odorless gas with the symbol O, essential for life on Earth. It makes up 21% of the atmosphere and is the third most abundant element in the universe. Oxygen is highly reactive and forms compounds with most other elements. It is used in industrial processes, medical treatments, and rocket propulsion. In its liquid and solid forms, oxygen is pale blue. All known forms of life depend on oxygen for cellular respiration and energy production.
Oxygen Fact Check
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
OSHA's Respiratory Protection Standard considers any atmosphere with an oxygen level below 19.5 percent to be oxygen-deficient and immediately dangerous to life or health.
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
An oxygen-deficient atmosphere is defined as one with an oxygen concentration below 19.5% by volume at sea level.
|
Phosgene (COCl2)
Phosgene is a colorless gas with a suffocating odor reminiscent of moldy hay. Its chemical formula is COCl2. Phosgene is highly toxic and was used as a chemical weapon during World War I. It causes severe irritation and damage to the lungs, leading to fluid buildup (pulmonary edema) and potentially death. Industrially, phosgene is used in the production of plastics, pesticides, and pharmaceuticals. Strict safety measures and emergency response plans are essential when working with this dangerous substance.
Phosgene Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.1 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.1 ppm average over 10 hours
Ceiling: 0.2 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 0.1 ppm average over 8 hours
|
Phosphine (PH₃)
Phosphine is a colorless, flammable, and highly toxic gas with a garlic-like odor. Its chemical formula is PH3. Phosphine is used in the semiconductor industry for doping silicon wafers and in the production of organophosphorus compounds. It is also used as a fumigant for stored grains and in the synthesis of flame retardants. Exposure to phosphine can cause respiratory irritation, pulmonary edema, and damage to the central nervous system. It can ignite spontaneously in air, requiring careful handling and storage.
Phosphine Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 0.3 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 0.3 ppm average over 10 hours
Ceiling: 1 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 0.3 ppm average over 8 hours
|
Propane (C3H8)
Propane (C3H8) is a colorless, odorless, and flammable hydrocarbon gas. It is commonly used as a fuel for heating, cooking, and powering vehicles. Propane is stored and transported as a liquid under pressure in tanks or cylinders. It is a clean-burning alternative to other fossil fuels, with wide-ranging applications in both residential and industrial settings.
Propane Fact Sheet.
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1000 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 1000 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 1000 ppm average over 8 hours
|
Silane (SiH4)
Silane is a colorless, flammable gas with the chemical formula SiH4. It has a sharp, repulsive odor and is lighter than air. Silane is used in the semiconductor industry for depositing thin films of silicon dioxide and silicon nitride. It is also used in the production of solar cells and as a reducing agent in organic synthesis. Silane is pyrophoric, meaning it can ignite spontaneously in air, and it forms explosive mixtures with air. Proper ventilation, leak detection, and fire suppression systems are crucial when handling silane.
Silane Fact Sheet
Agency
|
Limits
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 5 ppm average over 10 hours
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 5 ppm average over 8 hours
|
Sulfur Dioxide (SO2)
Sulfur dioxide is a colorless, pungent gas with the chemical formula SO2. It is formed during the combustion of sulfur-containing fuels and is a major air pollutant. Sulfur dioxide contributes to acid rain and can cause respiratory irritation, aggravating asthma and other lung conditions. In the food industry, it is used as a preservative in dried fruits and wine. Sulfur dioxide is also used in the production of sulfuric acid and as a bleaching agent for paper and textiles. Exposure to high concentrations can be life-threatening.
Sulfur Dioxide Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 5 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 2 ppm average over 10 hours
Ceiling: 5 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 0.25 ppm average over 8 hours
|
|
World Health Organization (WHO)
|
AQG: 40 µg/m3 average over 24 hours |
|
Environmental Protection Agency (EPA)
|
NAAQS: 7 ppb average over 1 hour
|
Styrene (C8H8)
Styrene is a colorless to yellowish, oily liquid with a sweet, floral odor. Its chemical formula is C6H5CH=CH2. Styrene is primarily used in the production of polystyrene plastics, rubber, and resins. It is also found in small amounts in natural sources like coffee and cinnamon. Styrene is highly flammable and can polymerize exothermically if not inhibited. Chronic exposure to styrene can cause neurological effects and increase the risk of certain cancers. Proper ventilation and personal protective equipment are necessary when working with this substance.
Styrene Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 100 ppm average over 8 hours
Ceiling: 200 ppm average over 15 minutes
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 50 ppm average over 10 hours
Ceiling: 100 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 20 ppm average over 8 hours
|
Vinyl Chloride (C2H3Cl)
Vinyl chloride is a colorless, flammable gas with a sweet odor. Its chemical formula is C2H3Cl, featuring a double bond between the two carbon atoms. Vinyl chloride is primarily used in the production of polyvinyl chloride (PVC), a widely used plastic material. It is also a key ingredient in the manufacturing of various plastics, adhesives, and coatings. Vinyl chloride is a known human carcinogen, and chronic exposure can lead to liver damage and a rare form of liver cancer called angiosarcoma. Strict regulations and safety measures are in place to minimize occupational exposure and environmental contamination.
Vinyl Chloride Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 1 ppm average over 8 hours
Ceiling: 5 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV: 1 ppm average over 8 hours
|
Xylenes (C6H4(CH3)2)
Xylenes are aromatic hydrocarbons consisting of a benzene ring with two methyl substituents. The three isomers are o-xylene, m-xylene, and p-xylene, differing in the positions of the methyl groups. Xylenes are colorless, flammable liquids with a sweet odor, commonly used as solvents and in the production of other chemicals.
Xylenes Fact Sheet
Agency
|
Limits
|
|
Occupational Safety & Health Administration (OSHA)
|
PEL: 100 ppm average over 8 hours
|
|
National Institute for Occupational Safety & Health (NIOSH)
|
REL: 100 ppm average over 10 hours
Ceiling: 150 ppm average over 15 minutes
|
|
American Conference of Government Industrial Hygienists (ACGIH)
|
TLV:
100 ppm average over 8 hours
STEL: 150 ppm average over 15 minutes
|
source
https://www.forensicsdetectors.com/blogs/articles/gas-exposure-limit-tables